Sun
Pharmaceutical Industries Antibiotic Drug Bactrim
Gave Young Girl Lung Failure That Left Her Fighting
For Her Life And Others Who Took It Needing Lung
Transplants
April 14. 2026
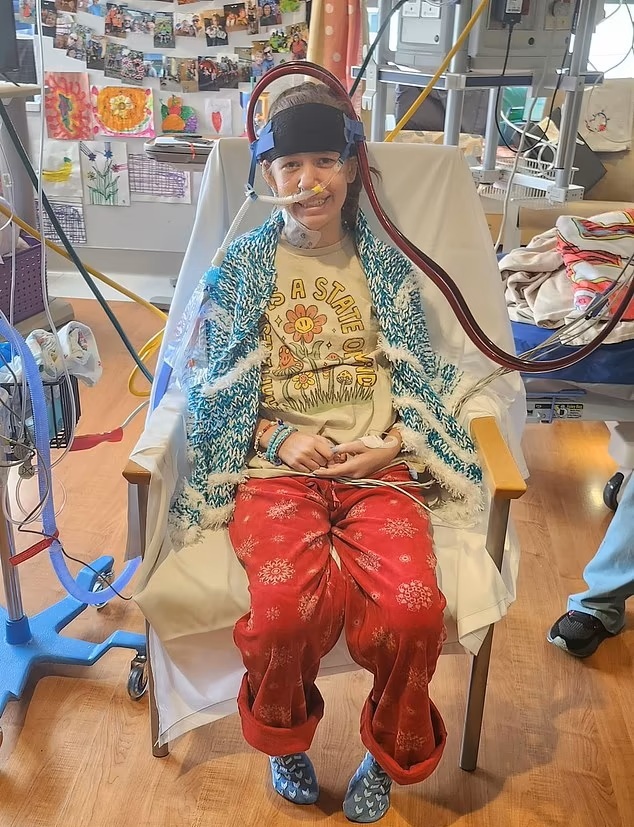
12-year-old Emmie Bellucci in the hospital after taking the
antibiotic Bactrim and it causing lung failure
This is another in the series of articles I've been writing
about the antibiotic Bactrim (trimethoprim/sulfamethoxazole), manufactured by
the company Sun Pharmaceutical Industries. Bactrim has killed over 300 people
0including children) in America and sickened many more. My mom was prescribed
Bactrim on August 31, 2023 and it greatly damaged her health. I hate that drug
for what it did to my mom's health. She nearly died after one week of taking it
as prescribed.
I've found other cases of Bactrim harming the public. The Daily
Mail reported in January 2025, "Doctors are sounding the alarm about a common
antibiotic taken by millions that is linked to a number of deaths and extreme
reactions. Taken by about 3million Americans every year,"

Sun Pharmaceutical Industries has been the subject of
several drug recalls due to safety issues
A number of children and some adults who were prescribed Bactrim
also suffered lung failure and needed transplants..12-year-old Emmie Bellucci
went into organ failure (lungs) after taking Bactrim. She was hospitalized for
5-months and a tracheostomy was performed to help her breathe.
Avoid Bactrim at all costs. It's a terrible drug. It's pretty
sad when one of the main side effects of a pharma drug is death. There are other
antibiotics that are better and more effective with far less side effects than
Bactrim (trimethoprim/sulfamethoxazole). Remember the name Bactrim and don't
take it.
STORY SOURCE
Doctors' warning over killer
antibiotic taken by millions that causes organ
failure
READ MORE: Antibiotic taken by millions is pulled
from shelves in Alabama
The Bellucci family is raising money via GoFundMe for
medical expenses.
11:59 EDT, 24 January 2025 | Updated: 17:22 EDT, 24
January 2025 - Doctors are sounding the alarm about a common antibiotic
taken by millions that is linked to a number of deaths and extreme
reactions. Taken by about 3million Americans every year, Bactrim is
usually used to treat a range of bacterial infections such as ear, chest
and stomach bugs, as well as UTIs.
Emmie Bellucci, 12, from Texas, was prescribed two courses
of the antibiotic for cystic acne - a severe form that causes painful
lumps deep in the skin - last year. A severe reaction caused her lungs to
become severely injured and shut down. She has been hospitalized with five
months, reliant on a tube in her windpipe - a tracheostomy - to help her
breathe. Doctors believe the drug triggers an immune response in young
patients like Emmie that causes the body to attacks healthy tissue that
lines lungs, causing respiratory failure. Experts investigating the rare
complication are now calling for increased research on Bactrim's long-term
harms and tests to look for risk factors before describing the drug...
https://www.dailymail.co.uk
My Journey of Discovery
In 2018, one of my long term ECMO patients was featured
in a CNN Health article as she walked and talked on ECMO. This article
caught the attention of other patients and their families who felt like
their stories were similar to our patient. They shared their medical
histories that often lead to severe lung failure requiring ECMO (lung
bypass), lung transplant, and/or death after Trimethoprim-Sulfamethoxazole
(Bactrim) exposure. Since then we have found 20 patients, published
multiple articles which are linked below and continue to actively evaluate
new patients for this disease. As such, there was a label change by the
FDA on package inserts in 2021.
https://www.jennamillermd.com/bactrim
Common Antibiotic Combination Causes Sudden Death
January 6, 2015 - Hyperkalemia may be a concern when
patients are prescribed trimethoprim/sulfamethoxazole. Now and again, a
study comes along that changes how health care professionals think about
prescribing. Recently, research released by the British Medical Journal
implicated trimethoprim/sulfamethoxazole as a cause for increased risk of
sudden death in specific populations.
This particular study gathered data for more than 17
years, from 1994 through 2012, and included patients aged 66 years or
older who were being treated with either an angiotensin-converting enzyme
(ACE) inhibitor or angiotensin receptor blocker (ARB) and then suddenly
died within 7 or 14 days of starting treatment as an outpatient with an
oral antibiotic. Of the more than 1.5 million patients included in the
study, nearly 40,000 died suddenly after beginning oral antibiotic
treatment. The results showed that, in this specific patient population, 3
of every 1000 individuals taking either ACE inhibitors or ARBs who then
begin treatment with trimethoprim/sulfamethoxazole will die suddenly
within 7 to 14 days.
Those results are significant when compared with the
control cohort, which recorded only 1 sudden death per 1000 patients.
Previous research has shown that older patients treated with trimethoprim/sulfamethoxazole
who are concurrently taking ACE inhibitors or ARBs have an increased risk
for hospitalization due to hyperkalemia. The authors of that prior study
theorized that such increased risk of sudden death in the cohort was
associated with the quick and clinically significant rise in potassium,
which then caused an unrecognized arrhythmic death.
The authors also implied that sudden death in those
patients might be inappropriately attributed to something other than
hyperkalemia, such as an underlying cardiovascular disease. With more than
250 million prescriptions for ACE inhibitors and ARBs annually, and more
than 20 million prescriptions for trimethoprim/sulfamethoxazole dispensed
each year in the United States, chances are that the 2 will be prescribed
together. When appropriate, it is prudent for clinicians to evaluate
alternative antibiotic regimens, consider a decreased duration of
treatment, or closely monitor serum potassium levels in this particular
patient population.
https://www.pharmacytimes.com
RELATED ARTICLES
Sun Pharmaceutical Industries
Factory Suspended By The Chinese Government After
Failing Several U.S. Government Safety Inspections
Confirming Previous Site Claims Their Drugs Are
Dangerous And Deadly
Sun Pharmaceutical Industries Is Not Doing What
Is In The Best Interest Of Their Shareholders Which Will Lead To Financial
And Legal Trouble
Sun Pharmaceutical Industries
Has A History Of Safety Violations At Their Plants That Produced The
Deadly Antibiotic Bactrim
Sun Pharmaceutical Industries Are Releasing
Dangerous And Deadly Drugs That Keep Getting Recalled In America And
Abroad
There Is Stock Fraud
Transpiring At Sun Pharmaceutical Industries
First Guaranty Mortgage
Financially Collapses Under Boycott
Cano Health Closes Locations After
Filing For Bankruptcy
Site
Exclusives